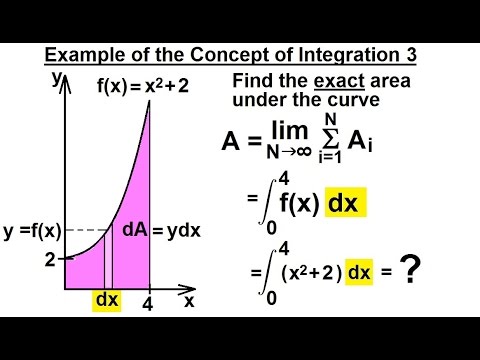
A data transformation: A boolean-condition can be used to change the state of points. (Since active points do not need to be in one region, we do not use the region of interest term here, but such region can be easy selected). Inactive points are excluded from fitting and all calculations. For calculating the area under the curve we divide the whole area in the form of few rectangular strips of height/length f(x 0 ) and breadth dx and the total area under the curve can be approximately obtained by adding the areas of all the rectangular strips. In Fityk, each point is either active or inactive. This fraction is normalized by multiplying by each dosage form's respective dose. Area under a curve yf(x) can be integrating the function between xa and xb. This is done by comparing the AUC of the non-intravenous dosage form with the AUC for the drug administered intravenously. oral tablet, suppository, subcutaneous, etc.) compared with the bioavailability of the same drug administered intravenously (IV). īioavailability can be measured in terms of "absolute bioavailability" or "relative bioavailability".Ībsolute bioavailability refers to the bioavailability of drug when administered via a non-intravenous (non-IV) dosage form (i.e. 
time plots are typically gathered using C-14 labeled drugs and AMS (accelerated mass spectrometry). In order to determine the respective AUCs, the serum concentration vs. This is often measured by quantifying the "AUC". In pharmacokinetics, bioavailability generally refers to the fraction of drug that is absorbed systemically and is thus available to produce a biological effect. The amount eliminated by the body (mass) = clearance (volume/time) * AUC (mass*time/volume). Also, AUC is referenced when talking about elimination. For example, gentamicin is an antibiotic that can be nephrotoxic (kidney damaging) and ototoxic (hearing damaging) measurement of gentamicin through concentrations in a patient's plasma and calculation of the AUC is used to guide the dosage of this drug.ĪUC becomes useful for knowing the average concentration over a time interval, AUC/t. darea(y) gives the area under interpolated data points. Another use is in the therapeutic drug monitoring of drugs with a narrow therapeutic index. Fityk is a program for nonlinear fitting of analytical functions. AUC is a useful metric when trying to determine whether two formulations of the same dose (for example a capsule and a tablet) result in equal amounts of tissue or plasma exposure. The AUC (from zero to infinity) represents the total drug exposure across time. 
Interpretation and usefulness of AUC values 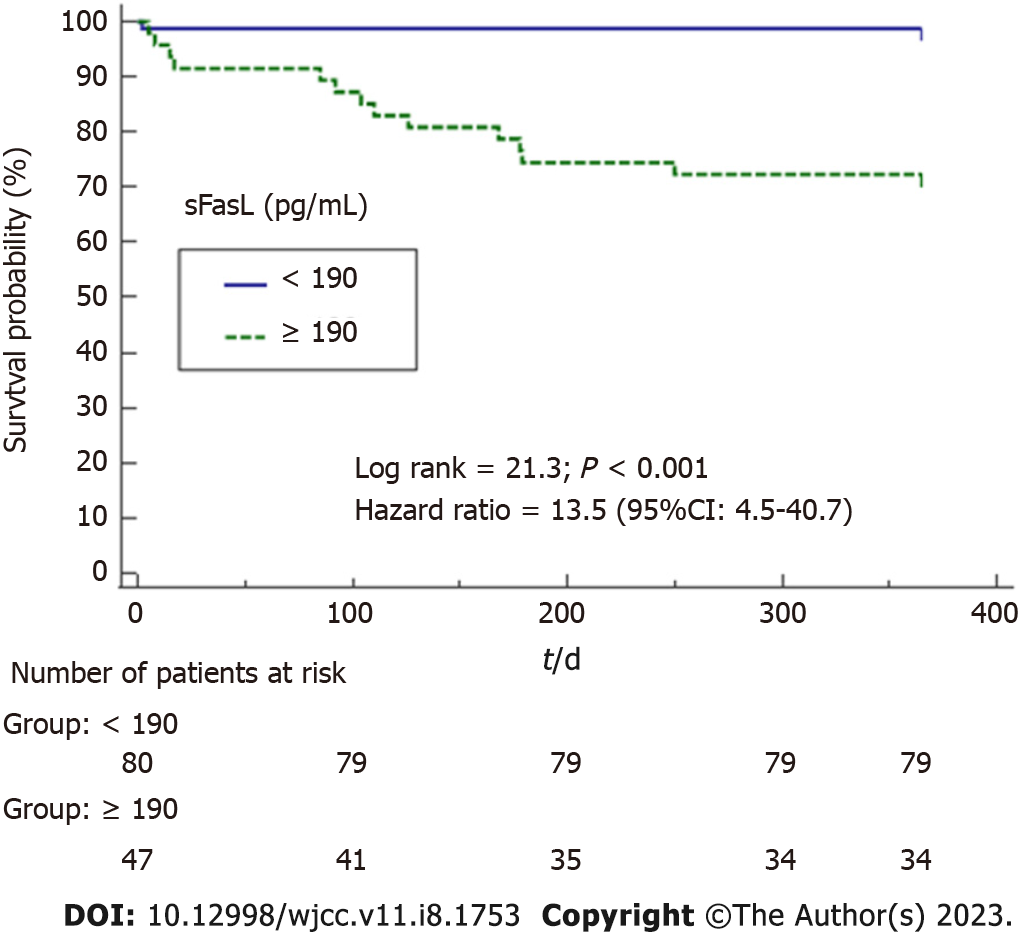
In practice, the drug concentration is measured at certain discrete points in time and the trapezoidal rule is used to estimate AUC. In the field of pharmacokinetics, the area under the curve ( AUC) is the definite integral of the concentration of a drug in blood plasma as a function of time (this can be done using liquid chromatography–mass spectrometry ). > To unsubscribe from this group and stop receiving emails from it, send an email to To view this discussion on the web visit. > You received this message because you are subscribed to the Google Groups "fityk-users" group. > Could you be more specific? What exactly means that it works not so well? > I have doubts about calculating the area under the curve of my dataset in certain ranges. > On Wed, at 4:54 PM Marcin Wojdyr wrote: The number I obtain from the peak at the right of 550 nm is roughly the same as the peak at the left of 600, (10.3731 and 14.643 respectively), when I was expecting at least three times larger.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
